Solvatomorphism and phase transformation of CL-20: probing properties and investigating mechanisms†
Abstract
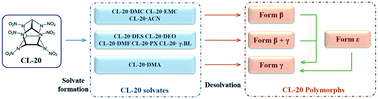
Five novel solvates of 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane (CL-20) were crystallized and their crystal structures were elucidated. Solvate formation was found to be a multi-point molecular recognition and self-assembly process through C–H⋯O synthons. Hirshfeld surface analysis confirms that numerous C–H⋯O interactions are formed between the host and guest molecules, and the C–H⋯O contribution is greater for solvates than for polymorphs. Thermal analysis was performed to evaluate the thermal stability and verify the stoichiometry of solvates. The phase transformation during the desolvation process was in situ monitored by variable temperature powder X-ray diffraction. Both structural similarity and Ostwald's rule of stages play important roles in determining the polymorphs obtained after desolvation. These findings provide a promising route for multi-component crystal design of CL-20 using crystal engineering strategies.


 Please wait while we load your content...
Please wait while we load your content...