Formamide-assisted one-step synthesis of BiOCOOH and Bi/BiOCOOH micro-/nanostructures with tunable morphologies and composition and their photocatalytic activities†
Abstract
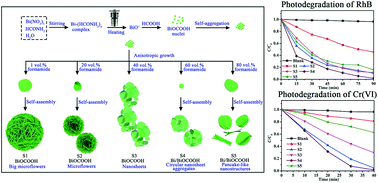
A series of BiOCOOH and Bi/BiOCOOH heterojunction micro-/nanostructures with various morphologies have been successfully synthesized by a one-step hydrothermal process with the assistance of formamide. The morphology, structure and composition of the as-obtained samples and the effect of formamide were systematically characterized and investigated. The results reveal that formamide plays multiple roles in the synthesis of the BiOCOOH and Bi/BiOCOOH heterojunctions, which acts as the carbon and alkali source. Moreover, the formic acid abundantly provided by the hydrolysis of formamide serves as the reductant and structure-directing agent. The photocatalytic activities of all the catalysts were evaluated by the degradation of rhodamine B (RhB) and reduction of potassium dichromate (K2Cr2O7) under simulated sunlight irradiation. It is found that the photocatalytic activity is closely related to the morphology and composition of the photocatalyst. The well-defined 3D flower-like BiOCOOH prepared at 20 vol% formamide shows a higher photocatalytic activity than the other BiOCOOH samples, which could be attributed to its hierarchical nanostructure, larger specific surface area and stronger adsorption capacity. Meanwhile, the Bi/BiOCOOH heterojunction obtained at 60 vol% formamide shows the best photocatalytic performance due to the increased light absorbance capacity and more efficient separation and transfer of photogenerated carriers with the existence of in situ deposited metallic Bi. Trapping experiments reveal that h+ and ˙O2− are the main active species in the photodegradation process and a possible photocatalytic mechanism is proposed.


 Please wait while we load your content...
Please wait while we load your content...