Two donor–acceptor (D–A) type Zn(ii) complexes as fluorescent probes for highly selective detection of iodide†
Abstract
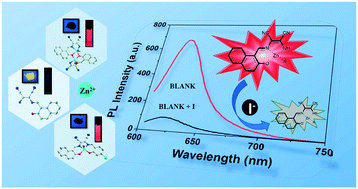
Two D–A type Zn(II) coordination complexes ([Zn2(L1)2(MeCN)(MeOH)]·(MeCN) (1) and [ZnL2(H2O)] (2)) were synthesized from diaminomaleonitrile-based (DMN) Schiff-base ligands (L1 = 2-amino-3-[(2-hydroxynaphthalen-1-ylmethylene)-amino]-but-2-enedinitrile, L2 = 2-[(4-chloro-2-hydroxybenzylidene)-amino]-3-[(2-hydroxynaphthalen-1-ylmethylene)-amino]-but-2-enedinitrile). Based on the weak blue fluorescence in THF solution and the bright yellow fluorescence in the solid state, the ligand L1 shows obvious aggregation induced emission (AIE) in a THF–H2O solvent system. The fluorescence emission of complexes 1 and 2 exhibits a significant red-shift relative to that of the ligand in THF solution. Interestingly, because of aggregation caused by the interaction between L1 and iodide (I−), complexes 1 and 2 exhibit excellent selectivity for fluorescence detecting iodide, and the quenching efficiency of 2 with iodide is higher than that of 1.


 Please wait while we load your content...
Please wait while we load your content...