The contributions of distinct Pd surface sites in palladium–ceria catalysts to low-temperature CO oxidation†
Abstract
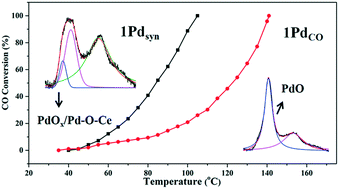
Palladium-loaded catalysts exhibit promising low-temperature catalytic activity toward CO oxidation. In this work, ceria with well-defined facets was utilized as a support to investigate the structure evolution of palladium species and, thereby, the correlation of this with the catalytic properties. A typical relationship between the distribution of palladium species and the Pd content and calcination conditions has been detected. Calcinating under an O2 atmosphere results in three different Pd species: PdOx aggregates, PdO nanoparticles, and Pd–O–Ce solid solution. The results show that high palladium-loading facilitates the enrichment of Pd in PdOx and PdO forms, while low palladium-loading induces the dissolution of considerable amounts of Pd into the bulk to form Pd–O–Ce. Further structural evolution resulting from the decomposition of PdOx to Pd–O–Ce has been detected during the calcination process under a CO atmosphere. Furthermore, it is found that calcinating in synthetic gas (a mixture of CO and O2) stimulates the regeneration of decomposed palladium species on the surface. In correlation with the structure evolution, PdOx and PdO species are essential for improving the low-temperature catalytic properties.


 Please wait while we load your content...
Please wait while we load your content...