Injection time controls the final morphology of nanocrystals during in situ-seeding synthesis of silver nanodisks†
Abstract
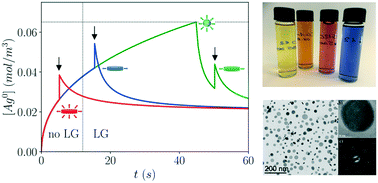
We investigate an in situ-seeding synthesis of silver nanodisks based on the sequential addition of weak and strong reducing agents, ascorbic acid and sodium borohydride respectively, to silver nitrate at room temperature and in the presence of polyvinylpyrrolidone. Well-defined silver nanodisks with a uniform thickness of 8 nm and a distribution of diameters around a mean of 22 nm can thus be prepared, the diameter being tunable. We report and explain the influence of important experimental control parameters on the quality of the nanoparticle dispersion synthesized. In particular, we show that a dispersion containing exclusively nanodisks can only be obtained if the second reducer is injected in a specific time window at room temperature. The rapid injection of concentrated borohydride triggers a burst nucleation creating seeds with structural defects. Seeds are then grown from silver atoms produced by the reduction of remaining silver ions by ascorbic acid. When the rate of production of these atoms is slow enough, seeds grow along the seed defects and only nanodisks are obtained. The ability to maintain a slow enough production rate after burst nucleation is thus essential. This critical reduction rate depends on the injection time as the latter determines the amount of reactants remaining after nucleation. The influence of temperature is subtle as it affects both reaction kinetics and adatom diffusion on the nanoparticle surfaces. Provided these subtle kinetic effects are understood and under control, the present in situ-seeding synthesis yields good quality nanodisk dispersions in only a few minutes. These results suggest realistic and robust pathways for a scalable production of silver nanodisks in continuous flow reactors most suited to industrial needs.


 Please wait while we load your content...
Please wait while we load your content...