Phase selection of calcium carbonate crystals under the induction of lignin monomer model compounds
Abstract
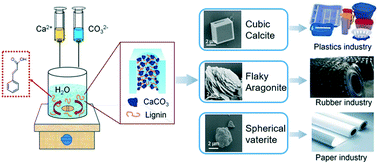
As an important class of natural resources, lignin and its utilization receive more and more attention. However, it is treated as waste in paper industry and causes serious pollution of natural water bodies. Meanwhile, the uncontrollable surface topography and the crystal form of natural calcium carbonate (CaCO3) severely restrict its application in the industrial field. Biomineralization may provide a perfect solution for these two issues. In this work, two types of specific crystal forms, cinnamic acid & aragonite crystal (CAAs) and cinnamic acid & vaterite crystal (CAVs) were directly synthesized in the presence of cinnamic acid. Two kinds of polymorph states were also found in the growth process of CaCO3. The structural properties of CAAs and CAVs as well as their polymorph states were characterized by FESEM, AFM, FTIR spectroscopy, XRD and TGA. The results showed that CAAs and CAVs held the crystal forms of aragonite and vaterite, respectively. The crystal forms of CaCO3 followed the growth rule that they dynamically changed from calcite to aragonite and then to vaterite. The formation mechanism is illustrated in the nucleation and crystal growth processes of CaCO3; cinnamic acid first formed various surfaces with the change in ambient conditions. These made CaCO3 to form different crystal forms and surface structures.


 Please wait while we load your content...
Please wait while we load your content...