Crystal formation of 1D coordination polymers based on chiral, achiral and racemic 1,2-cyclohexane scaffolds†
Abstract
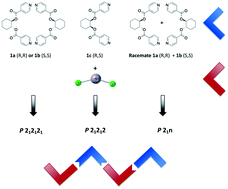
Enantiomerically pure ligands (1a and 1b) and meso1c based on trans-1,2-cyclohexanediol and cis-1,2-cyclohexanediol, respectively, were used for the formation of zigzag 1D coordination polymers, when combined with bent HgCl2. A racemic mixture or 1a/1b in a 1/1 ratio has also been studied. Four 1D coordination polymers have been obtained and their structures have been analysed using X-ray diffraction on single crystals and also on powder. 1a-HgCl2 (and isostructural 1b-HgCl2) crystallizes in the non-centrosymmetric P212121 space group, 1c-HgCl2 in the non-centrosymmetric P21212 space group and 1a0.51b0.5-HgCl2 crystallises in the centrosymmetric P21/n space group. The analysis of network energies, evaluated using a partial charges calculation method (PACHA) used here for the first time for 1D coordination networks, allows evaluation of energetical parameters like the packing energy (PE) of each crystalline compound, as well as their cooperativity index (CI), illustrating the influence of chirality on the packing of this series of 1D coordination polymers. It has been shown that the use of the achiral ligand 1c requires the highest cooperativity between chains.
- This article is part of the themed collection: Introducing the CrystEngComm Advisory Board and their research


 Please wait while we load your content...
Please wait while we load your content...