Simple one-pot synthesis of Cu4SnS4 nanoplates and temperature-induced phase transformation mechanism†
Abstract
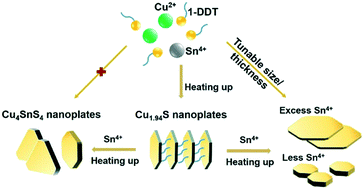
A simple one-pot heat-up method has been developed to synthesize Cu4SnS4 nanoplates with highly exposed (002) surfaces. Temperature-dependent phase transformation from monoclinic Cu1.94S to orthorhombic Cu4SnS4, and eventually to partially triclinic Cu2SnS3 has been observed. It is revealed that the Sn4+ existing in the laminar structure of Cu1.94S nanoplates starts being incorporated into the Cu1.94S crystal structure at a temperature of around 220 °C, and form pure orthorhombic Cu4SnS4 nanoplates at 260 °C. As the orthorhombic structure of Cu4SnS4 is metastable, it undergoes a phase transformation to the more stable Cu2SnS3 phase at a higher reaction temperature of 270 °C. Furthermore, the size and thickness of Cu4SnS4 nanoplates can be well tuned by the amount of Sn content in the initial reaction mixture to form Cu1.94S nanoplates. Additionally, the formation of Cu1.94S nanoplates is essential for the formation of pure Cu4SnS4 nanocrystals at a fixed reaction temperature. UV-vis and Mott–Schottky analysis show that the as-prepared orthorhombic Cu4SnS4 nanoplates exhibit n-type semiconducting behavior with a band gap of about 1.65 eV. The valence and conduction band potentials have also been determined via theoretical calculations. We believe that our synthetic method and findings will be attractive for the synthesis of other copper-based chalcogenide nanoplates as well as their potential catalytic applications.


 Please wait while we load your content...
Please wait while we load your content...
