Five naphthalene-amide-bridged Ni(ii) complexes: electrochemistry, bifunctional fluorescence responses, removal of contaminants and optimization by CVD†
Abstract
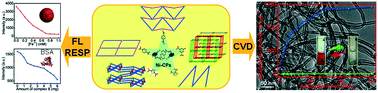
To research the effect of carboxylates on the assembly of Ni(II) coordination polymers, a naphthalene-amide ligand L (N,N′-bis(pyridin-3-ylmethyl)naphthalene-2,6-dicarboxamide) and five dicarboxylates [1,4-H2BDC = 1,4-benzenedicarboxylic acid, 1,4-H2PDC = 1,4-phenyldiacetic acid, 5-H2HIP = 5-hydroxyisophthalic acid, 3,4-H2TDC = 3,4-thiophenedicarboxylic acid, 2,5-H2TDC = 2,5-thiophenedicarboxylic acid] were selected to combine with Ni(II) under hydrothermal conditions. Five coordination polymers [Ni(L)(1,4-BDC)(H2O)2] (1), [Ni(L)(1,4-PDC)(H2O)2] (2), [Ni(L)(5-HIP)(H2O)2] (3), [Ni(L)(3,4-TDC)(H2O)2]·3H2O (4) and [Ni(L)(2,5-TDC)(H2O)]2·3H2O (5) were synthesized and structurally characterized by IR, PXRD and X-ray single-crystal diffraction. The title complexes show various structures: 2D 4-connected sql nets (1 and 2), a 3D cds framework of 3, 1D zigzag chain of 4 and 3D irl framework of 5. The electrochemical behaviors of 1–5 and the solid-state luminescence and fluorescence responses of 5 to Fe3+ and bovine serum albumin (BSA) were studied. Encouraged by functional thiophene components, 4 and 5 were used as catalyst precursors to synthesize carbon nanotubes (CNT-4 and CNT-5) by the chemical vapor deposition (CVD) method and characterized by XPS, SEM, TEM and Raman spectroscopy. The removals of contaminants (rhodamine B and Congo red) by 4 and 5 and their derivatives CNT-4 and CNT-5 in the aqueous phase were also comparatively investigated.


 Please wait while we load your content...
Please wait while we load your content...