Mg0.8Zn0.2O microspheres: preparation, characterization and application for degrading organic dyes†
Abstract
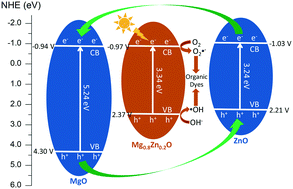
Photocatalytic degradation of organic pollutants in wastewater is one of the most promising strategies for environmental remediation, and photocatalysts as the prerequisite have received considerable attention. Herein we reported highly efficient, nontoxic, and inexpensive Mg0.8Zn0.2O microspheres with a diameter of around 36 μm prepared through reaction between CO32−, Mg2+ and Zn2+ for degrading organic dyes. The molar ratio between Mg2+ and Zn2+ was found to play a crucial role in determining the morphology and photocatalytic performance of the resulting product. According to time-dependent experiments, the plausible formation mechanism for the spherical-like Mg0.8Zn0.2O composite was proposed, in which irregular agglomerates composed of fine powders were first formed followed by their transformation into rod-like structures. Due to their thermal instability, the rod-like particles underwent further self-assembly and became a more stable spherical-like product with ZnCO3 embedded in the space between Mg5(CO3)4(OH)2·4H2O sheets. After investigating the photocatalytic performance, the results demonstrated that the developed Mg0.8Zn0.2O composite was not only superior to other state-of-the-art catalysts such as P25 TiO2 and g-C3N4 in degrading organic dyes upon irradiation under either UV light or visible light, but also was an amphoteric photocatalyst more favorable for degrading organic dyes in both strongly acidic (pH 2–4) and strongly alkaline (pH 12) solutions. Moreover, its catalytic efficiency remained as high as 98.3% after six cycles. These features made the developed Mg0.8Zn0.2O composite promising in practical utilization for treatment of organic dyes. Further experiments including photoluminescence, adsorption and photoelectrochemical measurements accounted for the underlying photocatalytic mechanism of the Mg0.8Zn0.2O composite for degradation of organic dyes.


 Please wait while we load your content...
Please wait while we load your content...