Investigating the effects of activating agent morphology on the porosity and related capacitance of nanoporous carbons†
Abstract
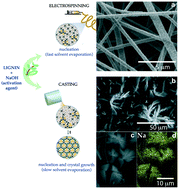
The increasing interest in nanostructured porous carbon materials in the field of energy storage and electrocatalysis has led to drastic improvement in their properties. These materials are still expensive to manufacture, often due to the harsh chemical treatments required to enhance porosity. In this article, we demonstrate that the process of activating carbons does not uniquely depend on the amount of activating agent (NaOH) but also on the contact area between the salt and the carbonaceous framework to be activated. We compare the microstructure, double layer capacitance and microporosity of carbon materials of the same chemical composition but obtained through two different protocols: electrospinning, which allows the formation of small NaOH domains (∼60 nm) homogeneously distributed across lignin fibres, and standard activation, which leads to the growth of larger NaOH domains within the lignin framework. As intimate contact between both phases ensures maximum activation, our electrospun material shows a remarkable capacitance (∼180 F g−1) in comparison to the material prepared through standard “mix and dry” activation procedure (∼10 F g−1). This is attributed to the generation of micropores (∼670 m2 g−1), tailored from the small activating salt template domains in intimate contact with the lignin framework. In summary, through this work we establish a relationship between the activating agent's morphology and its effect in porosity and energy storage performance. We present a facile method to interpenetrate the chemical activating agent within the carbon precursor structure before pyrolysis, through the efficient usage of the activating agent.
- This article is part of the themed collection: Crystal engineering for electrochemical applications


 Please wait while we load your content...
Please wait while we load your content...