A new series of lanthanide-based complexes with a bis(hydroxy)benzoxaborolone ligand: synthesis, crystal structure, and magnetic and optical properties†
Abstract
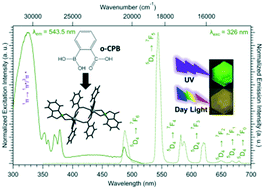
Reactions, under hydrothermal conditions, between lanthanide chlorides and the sodium salt of 2-carboxyphenylboronic acid lead to a series of lanthanide-based complexes with general chemical formula [Ln2(C7H5O2)4(C7O4H6B)2·4H2O] with Ln = Eu–Dy. During the synthetic process, the 2-carboxyphenylboronate ligand decomposes leading to benzoate (C7H5O2) and bis(hydroxy)benzoxaborolone (C7O2H6B). The crystal structure was solved on the basis of the Tb-derivative. It crystalizes in the triclinic system, space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) (n° 2) with a = 9.3329(13) Å, b = 16.012(2) Å, c = 16.212(2) Å, α = 107.789(5)°, β = 90.226(5)°, γ = 97.585(5)°, V = 2284.16(52) Å3 and Z = 2. Its crystal structure can be described on the basis of di-lanthanides entities surrounded by ligands. Compounds that constitute this series are thermally stable up to 190 °C. Solid-state luminescence properties have been studied in detail. The magnetic properties of the Dy-derivative have also been explored. It exhibits single molecule magnet behavior. Single crystals of the sodium salt of 2-carboxyphenylboronic acid with chemical formula [Na(C7O4H6B)(H2O)]∞ have also been obtained during this study. This compound crystallizes in the orthorhombic system, space group P212121 (n° 19) with a = 4.5881(13) Å, b = 6.5905(16) Å, c = 29.644(8) Å, V = 897.0(4) Å3 and Z = 4. Its crystal structure is 2D and can be described as zigzag chains of sodium 2-carboxyphenylboronate bound to each other by coordination water molecules.
(n° 2) with a = 9.3329(13) Å, b = 16.012(2) Å, c = 16.212(2) Å, α = 107.789(5)°, β = 90.226(5)°, γ = 97.585(5)°, V = 2284.16(52) Å3 and Z = 2. Its crystal structure can be described on the basis of di-lanthanides entities surrounded by ligands. Compounds that constitute this series are thermally stable up to 190 °C. Solid-state luminescence properties have been studied in detail. The magnetic properties of the Dy-derivative have also been explored. It exhibits single molecule magnet behavior. Single crystals of the sodium salt of 2-carboxyphenylboronic acid with chemical formula [Na(C7O4H6B)(H2O)]∞ have also been obtained during this study. This compound crystallizes in the orthorhombic system, space group P212121 (n° 19) with a = 4.5881(13) Å, b = 6.5905(16) Å, c = 29.644(8) Å, V = 897.0(4) Å3 and Z = 4. Its crystal structure is 2D and can be described as zigzag chains of sodium 2-carboxyphenylboronate bound to each other by coordination water molecules.


 Please wait while we load your content...
Please wait while we load your content...