Seeking a novel energetic co-crystal strategy through the interfacial self-assembly of CL-20 and HMX nanocrystals†
Abstract
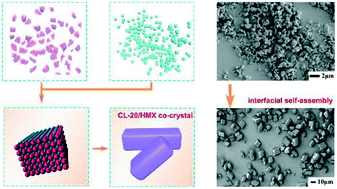
Energetic co-crystallization has been extensively explored as an effective strategy to balance the energy and sensitivity of energetic materials. In the present study, CL-20/HMX co-crystals with high purity, uniform morphology, well-proportioned size distribution, compact internal structure and reduced sensitivity were fabricated by a solvent-induced self-assembling approach using corresponding nanoparticles as the basic units. Such a CL-20/HMX co-crystal based on the self-assembly of a nano-explosive has been reported for the first time, providing a novel, high-yield, general and easily scaled-up method for the fabrication of energetic co-crystals. Notably, the formation processes of the as-synthesized co-crystals were determined as interfacial self-assembly at nanoparticle scale instead of the complete dissolution and nucleation process at the molecular level as observed with the traditional recrystallization technique. The completely formed CL-20/HMX co-crystal structure with few separate-assembled CL-20 or HMX crystals was charecterized by PXRD, TG-DSC, Raman, FT-IR, DG and SCXRD analyses. Additionally, the formation of intermolecular hydrogen bonds was confirmed by Terahertz (THz) analysis. The typical Avrami equation was constructed for modeling the kinetics of the assembly of CL-20/HMX co-crystals, and the self-assembly process can be generally summarized as the induction of nano-particles, oriented aggregation, surface integration and single co-crystal formation.


 Please wait while we load your content...
Please wait while we load your content...