Controllable synthesis of a 3D ZnS@MoO3 heterojunction via a hydrothermal method towards efficient NO purification under visible light†
Abstract
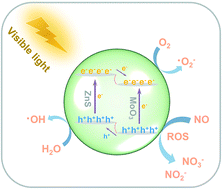
Heterojunctions can deliver superior photocatalytic efficiency via modulating the surface-interface reaction, enhancing light absorption and hindering charge carrier recombination. In this work, a 3D ZnS@MoO3 heterojunction was successfully synthesized by employing 3D sheet-like ZnO microspheres as a supporting matrix via a facile hydrothermal method, and the transformation of the ternary heterojunction (ZnO/ZnS/MoO3) to a binary heterojunction (ZnS@MoS2) was achieved by simple tuning of the ratios of the reactants in the initial system. A possible synthetic process is proposed. Photocatalytic experiments were performed to show that the as-prepared catalysts can effectively remove NO at a parts per billion (ppb) level, and the ZnS@MoO3 nanohybrids exhibited optimal oxidation ability that can be attributed to the suitable band alignment and interface effect, which can facilitate the spatial separation and transfer of photogenerated electron–hole pairs. Additionally, the possible mechanism and reaction pathway of NO purification are discussed through carefully designed experimental detection. Specifically, the results of the electron spin resonance trapping (ESR) spectra indicated that ˙O2− and ˙OH act as reactive oxygen species (ROS) and are responsible for the oxidation process in NO. More importantly, the promotion mechanism of the photocatalytic NO oxidation process is proposed by applying in situ diffuse reflectance infrared Fourier-transform spectroscopy (DRIFTS) to dynamically monitor and analyse the whole reaction process. Therefore, this work can provide new opportunities in designing efficient visible-light-driven heterojunction photocatalysts for NO oxidation.


 Please wait while we load your content...
Please wait while we load your content...