Leaching mechanism of bioapatite in carbonate-saturated water
Abstract
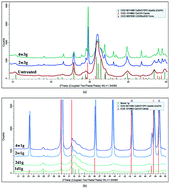
Bioapatite, the main inorganic component of bone, is similar to hydroxyapatite (HAp, Ca5[PO4]3(OH)) having some [CO3]2− content which plays an important role in leaching/precipitation processes in many biological lesions. We studied the leaching of powdered bovine tibia in soda-saturated (nahcolite, NaHCO3) water as a function of concentrations and treating periods. The reaction products were characterized by X-ray (XRD) and electron diffraction (SAED) techniques as well as high resolution transmission electron microscopy (HRTEM) and energy dispersive spectroscopy (EDS). We found evidence for dissolution of bone and precipitation of Ca-carbonate. The rate of carbonate precipitation was found to be a function of both the mass ratio of nahcolite/bone and the leaching time. According to XRD, calcite formed as a new crystalline phase at the expense of bioapatite. Based on HRTEM images, residual nanocrystalline apatite and unstable Ca-carbonate were also recognised, allowing the reconstruction of the intermediate stages of the leaching process. We propose a reaction sequence for the transformations of bioapatite in carbonated, alkaline media that may contribute to the understanding of the lesion processes of bone.


 Please wait while we load your content...
Please wait while we load your content...