Isoacenofuran: a novel quinoidal building block for efficient access to high-ordered polyacene derivatives†
Abstract
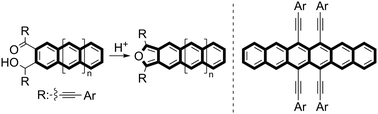
Herein, a simple and practical method for generating isoacenofuran, a new π-extended quinoidal building block, was developed. A three-step protocol involving double nucleophilic additions of alkynyllithiums to acene-2,3-dicarbaldehyde, mono-oxidation, and acid-promoted cyclization enables the generation of the target molecule, which is trapped by a dienophile to produce highly condensed acenequinones. Further transformations by double nucleophilic additions of alkynyllithium to hexacenequinone, followed by reductive aromatization, produce tetraalkynylhexacenes with a remarkably higher stability than that of the previously reported substituted hexacenes.


 Please wait while we load your content...
Please wait while we load your content...