Nickel-catalyzed insertions of vinylidenes into Si–H bonds†
Abstract
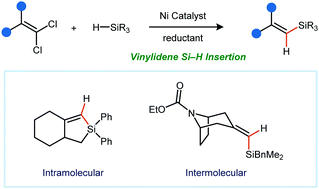
A nickel-catalyzed reductive cyclization of 1,1-dichloroalkenyl silanes is reported. The products of this reaction are unsaturated five- or six-membered silacycles. Intermolecular variants are also described, providing access to trisubstituted vinyl silanes that are not accessible by alkyne hydrosilylation or sila-Heck-type processes. A variety of silanes can be utilized, including those that serve as nucleophilic partners in Hiyama cross-coupling reactions. Mechanistic studies using deuterium-labelled silanes are described.
- This article is part of the themed collection: 2020 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...