Ionicity-dependent proton-coupled electron transfer of supramolecular self-assembled electroactive heterocycles†
Abstract
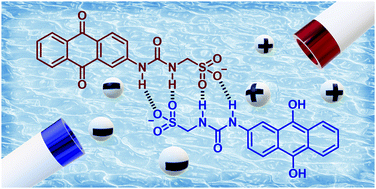
Herein, we investigate the electrochemical properties of a class of Supramolecular Self-associated Amphiphilic salts (SSAs). We show that varying ionic strength of an SSA solution can cause a switching of the thermodynamics and kinetics of electron transfer. The effect of self-assembly on proton-coupled electron transfer has implications for the understanding of electron transfer kinetics in aqueous organic redox flow batteries, especially at high concentration where organic–organic intermolecular interactions become dominant even for highly soluble organic species.


 Please wait while we load your content...
Please wait while we load your content...