A tetranuclear nickel cluster isolated in multiple high-valent states†
Abstract
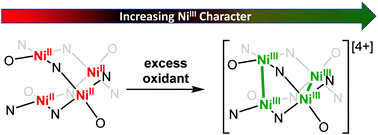
We report a series of high-valent tetranuclear nickel clusters isolated from the chemical oxidation of an all Ni(II) ([Ni4]) neutral cluster. Electrochemical analysis of [Ni4] reveals three reversible sequential oxidations at 0.248 V (1e−), 0.678 V (1e−), and 0.991 V (2e−) vs. Fc/Fc+ corresponding to mono-, di-, and tetra-oxidized species, [Ni4]+, [Ni4]2+, [Ni4]4+, respectively. Using spectroscopic, crystallographic, magnetometric, and computational techniques, we assign the primary loci of oxidations to the Ni centers in each case, thus resulting in the isolation of the first tetranuclear all-Ni(III) cluster, [Ni4]4+.
- This article is part of the themed collection: 2020 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...