Cobalt-catalyzed 2-(1-methylhydrazinyl)pyridine-assisted cyclization of thiophene-2-carbohydrazides with maleimides: efficient synthesis of thiophene-fused pyridones†
Abstract
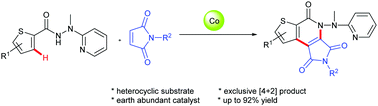
A cobalt-catalyzed direct C–H/N–H functionalization of thiophene-2-carbohydrazides with maleimides by utilizing 2-(1-methylhydrazinyl)pyridine (MHP) as an easily removable bidentate directing group has been developed. This formal [4+2] cycloaddition has been achieved for the first time, and it provides an alternative and versatile approach to construct thiophene-fused pyridones using an inexpensive cobalt catalyst. The C–H/N–H activation cascade protocol showed a high efficiency and a broad substrate scope, and the products were obtained in good to excellent yields.


 Please wait while we load your content...
Please wait while we load your content...