CH4 conversion over Ni/HZSM-5 catalyst in the absence of oxygen: decomposition or dehydroaromatization?†‡
Abstract
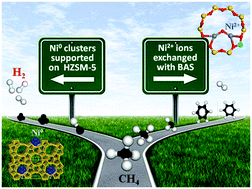
The different chemical states and locations of Ni species in HZSM-5 result in different directions of CH4 reaction. Supported Ni clusters caused complete CH4 decomposition, while Ni-exchanged sites anchored at Brønsted acid sites, can appropriately activate CH4 into CHx species which are important precursors to produce aromatics. This study suggests a catalyst construction strategy for selective conversion of CH4 towards value-added chemicals by placing active metal at specific sites.


 Please wait while we load your content...
Please wait while we load your content...