Photochemical α-carboxyalkylation of tryptophols and tryptamines via C–H functionalization†
Abstract
A process for the α-carboxyalkylation of tryptophols and tryptamines by the functionalization of C–H bonds under visible light irradiation has been developed. The photochemical strategy activated the C–Br bonds of α-bromo-alkylcarboxylic esters to provide carbon-centered radicals under the catalysis of Ir(III) photocatalyst and coupled with indole derivatives. This methodology displayed wide functional group tolerance and excellent regioselectivity, and was applied to the late-stage functionalization and preparation of indole-containing hybrids.
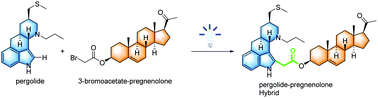


 Please wait while we load your content...
Please wait while we load your content...