Rational design of a mesoporous silica-based cathode for efficient trapping of polysulfides in Li–S batteries†
Abstract
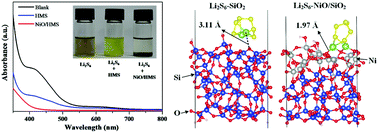
Lithium–sulfur batteries are one of the most promising candidates for next-generation energy storage systems. The major challenge hindering their commercialization is the polysulfide shuttle effect, which causes a series of problems including the loss of active materials, corrosion of the lithium anode, low coulombic efficiency, and poor cycling performance. In this work, we develop a mesoporous silica-based cathode for efficient trapping of lithium polysulfides (LiPSs). This cathode material consists of mesoporous silica (HMS), highly dispersed NiO nanoparticles embedded in the silica structure, and a conductive polymer (polypyrrole-ppy) prepared by in situ polymerization. We employ the concept of both the physical and chemical entrapment of LiPSs, i.e., physically trapping LiPSs by spatial confinement of LiPSs in the silica porous structure and physical adsorption of LiPSs by the silica surface, and chemically binding LiPSs by highly dispersed NiO nanoparticles in the silica structure. The NiO/silica/ppy/S cathode exhibits good cycling stability and maintains over 700 mA h g−1 after 300 cycles. As far as we know, this is the first time that mesoporous silica has been directly employed as a sulfur host material, rather than an additive. The present study opens up a window for nanoporous silica to be employed as the sulfur host.


 Please wait while we load your content...
Please wait while we load your content...