Disrupted intracellular redox balance with enhanced ROS generation and sensitive drug release for cancer therapy†
Abstract
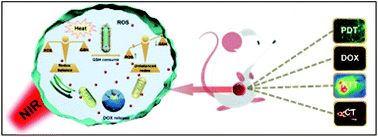
In this paper, a nanocomposite was constructed to achieve improved photodynamic therapy (PDT) via disrupting the redox balance in cancer cells. Firstly, Sb2Se3 nanorods were synthesized as a new photosensitizer, displaying high photothermal conversion efficiency (45.2%) and reactive oxygen species (ROS) production due to the narrow band gap (1.1 eV) and a good NIR response. Moreover, the mechanism was investigated, demonstrating that dissolved O2 and photoinduced electrons manipulated ROS generation. Then, mesoporous silica was coated outside to improve the biocompatibility and to supply abundant space for the anticancer drug (doxorubicin, DOX). The sensitive Se–Se linker was grafted outside via a silane coupling reaction to block DOX molecules in the mesopores. As we know, the Se–Se group is sensitive to GSH, which can induce Se–Se linker bond breakage and targeted drug release due to the high expression of GSH in tumor cells. What is more, the consumption of intracellular GSH can also disrupt the redox balance in cancer cells, which would promote the PDT efficiency. The high-Z element of Sb possesses a high X-ray attenuation coefficient, giving the composite high contrast in CT imaging. This is associated with thermal imaging and multi-therapy (PDT/PTT/chemotherapy) to reveal the potential application to cancer treatment.

 Please wait while we load your content...
Please wait while we load your content...