Synthesis of cyclic graft polymeric prodrugs with heterogeneous grafts of hydrophilic OEG and reducibly conjugated CPT for controlled release†
Abstract
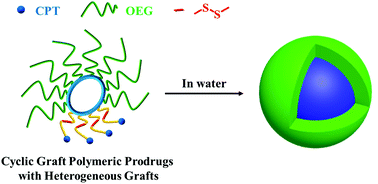
Fabrication of cyclic graft (cg) copolymer-based polymeric prodrugs by conjugation of drug molecules to cg copolymers via a dynamic covalent bond capable of responding to biorelevant signals integrates simultaneously the merits of cg copolymers and polymeric prodrugs for enhanced stability of nanocarriers and precise modulation of drug release kinetics. To completely eliminate the compromised drug conjugation efficiency due to the steric hindrance of hydrophilic grafts, it will be useful to develop cg polymeric prodrugs with heterogeneous grafts composed of hydrophilic polymers and drug species, respectively. For this purpose, we reported in this study the synthesis of cyclic graft polymeric prodrugs with heterogeneous grafts of hydrophilic oligo (ethylene glycol) (OEG) and reducibly conjugated camptothecin (CPT), cg-poly(oligo(ethylene glycol) monomethyl ether methacrylate)-b-poly((2-hydroxyethyl methacrylate)-disulfide link-camptothecin) (cg-P(OEGMA)-b-P(HEMA-SS-CPT), cg-prodrugs), via an integrated strategy of a previously reported diblock copolymer-based template and post-polymerization intermolecular click conjugation of a reducible CPT prodrug. The micelles self-assembled from cg-prodrugs on one hand had sufficient salt stability due to the branched cg structure, and on the other hand showed a reduction-triggered cleavage of the disulfide link for a promoted CPT release. Most importantly, we uncovered two interesting phenomena of the cg-based polymeric prodrugs as delivery vehicles: (i) the dimensions of both self-assemblies formed by the cg and bottlegraft (bg) polymers depend substantially on the molecular size of the cg and bg polymers likely due to the steric hindrance of the grafted structures of the cg and bg molecules and relatively low aggregation number of the self-assembled structures, and (ii) cg-prodrug-based micelles exhibited greater in vitro cytotoxicity against cancer cells despite the lower drug loading content (DLC) than the bg-based analogues, which results primarily from the faster reduction-triggered degradation and drug release as well as the greater cellular uptake efficiency of the former micelle prodrugs. Taken together, the developed cg-prodrugs provide great potential for chemotherapy, and the aforementioned interesting results will definitely inspire more upcoming studies on the future design and development of novel cg polymers for biomedical applications.
- This article is part of the themed collection: Biomaterials Science Emerging Investigators 2021


 Please wait while we load your content...
Please wait while we load your content...