Surface grafting of Fc-binding peptides as a simple platform to immobilize and identify antibodies that selectively capture circulating endothelial progenitor cells†
Abstract
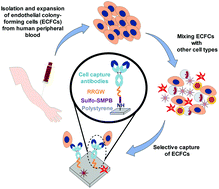
Antibody surface immobilization is a promising strategy to capture cells of interest from circulating fluids in vitro and in vivo. An application of particular interest in vascular interventions is to capture endothelial progenitor cells (EPCs) on the surface of stents to accelerate endothelialization. The clinical impact of EPC capture stents has been limited by the lack of efficient selective cell capture. Here, we describe a simple method to immobilize a variety of immunoglobulin G antibodies through their fragment crystallizable (Fc) regions via surface-conjugated RRGW peptides for cell capture applications. As an EPC capture model, peripheral blood endothelial colony-forming cells suspended in cell culture medium with up to 70% serum were captured by immobilized anti-CD144, anti-CD34 or anti-CD309 antibodies under laminar flow. The endothelial colony-forming cells were successfully enriched from a mixture with peripheral blood mononuclear cells using surfaces with anti-CD309 but not anti-CD45. This antibody immobilization approach holds great promise to engineer vascular biomaterials with improved EPC capture potential. The ease of immobilizing different antibodies using the same Fc-binding peptide surface grafting chemistry renders this platform suitable to screen antibodies that maximize cell capture efficiency and selectivity.


 Please wait while we load your content...
Please wait while we load your content...