Cholesterol moieties as building blocks for assembling nanoparticles to achieve effective oral delivery of insulin†
Abstract
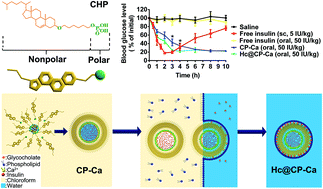
Although nanoparticles (NPs) have emerged as a promising strategy to improve oral absorption of insulin, most of them still have minimal oral bioavailability due to the harsh gastrointestinal (GI) environment and epithelial barriers. A novel platform of oral insulin delivery is herein proposed based on amphiphilic cholesterol-phosphate conjugate (CHP). The CHP was synthesized with a cholesterol moiety as a hydrophobic block and a phosphate group as a hydrophilic block. The structural characteristics of CHP made cholesteryl moieties to point outward to form a shell coated on the surface of NPs, and phosphate groups toward inside as the core via the water-in-oil (W/O) emulsion method. The additional Ca2+ could chelate to phosphate groups in the core to bind adjacent phosphate groups and further increased the stability of NPs (CP-Ca NPs). Phospholipid and glycocholate were modified on the surface of CP-Ca NPs to form hydrophilic corona coated CP-Ca NPs (Hc@CP-Ca NPs) and didn't change the structure of CP-Ca NPs. The NPs exhibited high encapsulation efficiency (>95%) and had high stability in the GI tract by reducing insulin release and protecting insulin from enzymatic degradation. Compared to free insulin, Hc@CP-Ca NPs demonstrated 31-fold higher cellular uptake in Caco-2 cells for 1 h incubation. Furthermore, the transepithelial transport efficiency of Hc@CP-Ca NPs was 12-fold higher than that of free insulin. Moreover, CP-Ca NPs and Hc@CP-Ca NPs generated a rapid-onset and long-lasting hypoglycemic effect following gavage in type 1 diabetic mellitus (T1DM) rats. The pharmacological activity and the relative bioavailability were increased to 13.5% and 12.2%, respectively. Thus, these results indicated that CHP assembled NPs may provide a promising potential for oral insulin delivery by protection and absorption promotion.


 Please wait while we load your content...
Please wait while we load your content...