A transport system based on a quantum dot-modified nanotracer is genetically and developmentally stable in pregnant mice†
Abstract
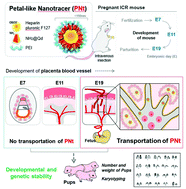
The use of nanoscale materials (NMs) could cause problems such as cytotoxicity, genomic aberration, and effects on human health, but the impacts of NM exposure during pregnancy remain uncharacterized in the context of clinical applications. It was sought to determine whether nanomaterials pass through the maternal–fetal junction at any stage of pregnancy. Quantum dots (QDs) coated with heparinized Pluronic 127 nanogels and polyethyleneimine (PEI) were administered to pregnant mice. The biodistribution of QDs, as well as their biological impacts on maternal and fetal health, was evaluated. Encapsulation of QDs with a nanogel coating produces a petal-like nanotracer (PNt), which could serve as a nano-carrier of genes or drugs. PNts were injected through the tail vein and accumulated in the liver, kidneys, and lungs. QD accumulation in reproductive organs (uterus, placenta, and fetus) differed among phases of pregnancy. In phase I (7 days of pregnancy), the QDs did not accumulate in the placenta or fetus, but by phase III (19 days) they had accumulated at high levels in both tissues. Karyotype analysis revealed that the PNt-treated pups did not have genetic abnormalities when dams were treated at any phase of pregnancy. PNts have the potential to serve as carriers of therapeutic agents for the treatment of the mother or fetus and these results have a significant impact on the development and application of QD-based NPs in pregnancy.


 Please wait while we load your content...
Please wait while we load your content...