ZBTB20-mediated titanium particle-induced peri-implant osteolysis by promoting macrophage inflammatory responses†
Abstract
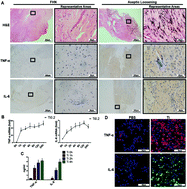
Aseptic loosening (AL) caused by wear particles released from implant surfaces is one of the main causes for the failure of artificial joints, which is initiated by macrophage inflammatory responses. Emerging evidence suggests that the member of a broad-complex, tramtrack, bric-a-brac/poxvirus and zinc finger (BTB/POZ) family as well as zinc finger and BTB domain-containing protein 20 (ZBTB20) can inhibit IκBα gene transcription, promote NF-κB activation, and initiate innate immune responses. The molecular mechanism(s) by which ZBTB20 contributes to titanium particle (TiP)-induced macrophage inflammatory responses and osteolysis has not been fully elucidated. Here, we showed that ZBTB20 increased either in the AL group's synovial membranes or in TiP-stimulated bone-marrow-derived macrophages (BMDMs) as compared to that in the control groups. Moreover, the knockdown of ZBTB20 led to the inhibition of proinflammatory factors induced by TiPs in BMDMs, such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interferon-β (IFN-β). Here, we also reported that the knockdown of ZBTB20 suppressed TiP-induced NF-κB activation and M1 polarization as well as stabilized the trans Golgi network (TGN) in BMDMs. The dual-luciferase reporter assay identified the binding between the IκBα promoter and ZBTB20, and IκBα knockdown could rescue the antiinflammatory effects induced by the ZBTB20 knockdown in BMDMs. Finally, we found that sh-ZBTB20 lentivirus injection could reduce TiP-induced osteolysis in mouse calvaria, inhibiting TiP-induced proinflammatory factors and loss of bone volume/total volume (BV/TV) as well as bone mineral density (BMD). These results suggest that ZBTB20 positively regulated NF-κB activation and M1 polarization as well as the production of TGN-derived tubular carriers in BMDMs, playing a positive role in macrophage activation and mouse cranial osteolysis induced by TiPs. It may be a potential therapeutic target for the prevention of aseptic loosening of prostheses.


 Please wait while we load your content...
Please wait while we load your content...