A photothermal-hypoxia sequentially activatable phase-change nanoagent for mitochondria-targeting tumor synergistic therapy†
Abstract
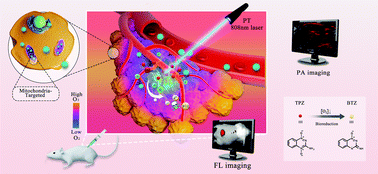
To enhance the specificity and efficiency of anti-tumor therapies, we have designed a multifunctional nanoparticle platform for photochemotherapy using fluorescence (FL) and photoacoustic (PA) imaging guidance. Nanoparticles (NPs) composed of a eutectic mixture of natural fatty acids that undergo a solid–liquid phase transition at 39 °C were used to encapsulate materials for the rapid and uniform release of the hypoxia-activated prodrug tirapazamine (TPZ) and the photosensitizer IR780, which targets the mitochondria of tumor cells and can be used to induce hypoxic cell death via photodynamic therapy and photothermal therapy. In vitro, the NPs containing TPZ and IR7890 exhibited appreciable cell uptake and triggered drug release when irradiated with a NIR laser. In vivo, photochemotherapy of the NPs achieved the best anti-tumor efficacy under PA and FL imaging guidance and monitoring. By combining IR780 mitochondria-targeting phototherapy with TPZ, we observed improved anti-tumor effectiveness and this has the potential to reduce the side effects of traditional chemotherapy. Herein, we demonstrate a new intracellular photochemotherapy nanosystem that co-encapsulates photosensitizers and hypoxia-activated drugs to enhance the overall anti-tumor effect precisely and efficiently.


 Please wait while we load your content...
Please wait while we load your content...