Hydrophilic titanium surfaces reduce neutrophil inflammatory response and NETosis†
Abstract
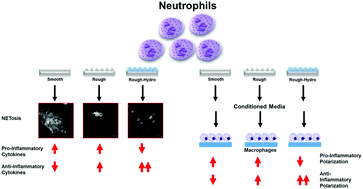
Biomaterial implantation triggers an immune response initially predominated by neutrophils, which activate an inflammatory cascade by producing cytokines, enzymes, immune cell recruitment chemokines, and DNA fiber networks called neutrophil extracellular traps (NETs). While the role of neutrophils has been studied extensively in infection, little is known of their role in the response to biomaterials, in this case titanium (Ti) implants. Furthermore, while implant surface modifications have been shown to attenuate pro-inflammatory polarization in other immune cells, their effects on neutrophil behavior is unknown. The aim of this study was to characterize the neutrophil response to Ti surface topography and hydrophilicity and understand how the products of biomaterial-induced neutrophil activation alters macrophage polarization. Murine neutrophils were isolated by density gradient centrifugation and plated on smooth, rough, and rough hydrophilic (rough-hydro) Ti surfaces. Neutrophils on rough-hydro Ti decreased pro-inflammatory cytokine and enzyme production as well as decreased NET formation compared to neutrophils on smooth and rough Ti. Conditioned media (CM) from neutrophils on smooth Ti enhanced pro-inflammatory macrophage polarization compared to CM from neutrophils on rough or rough-hydro Ti; pretreatment of neutrophils with a pharmacological NETosis inhibitor impaired this macrophage stimulation. Finally, co-culture of neutrophils and macrophages on Ti surfaces induced pro-inflammatory macrophage polarization compared to macrophages alone on surfaces, but this effect was ablated when neutrophils were pretreated with the NETosis inhibitor. These findings demonstrate that neutrophils are sensitive to changes in biomaterial surface properties and exhibit differential activation in response to Ti surface cues. Additionally, inhibition of NETosis enhanced anti-inflammatory macrophage polarization, suggesting NETosis as a possible therapeutic target for enhancing implant integration.


 Please wait while we load your content...
Please wait while we load your content...