Chain conformation transition induced host–guest assembly between triple helical curdlan and β-CD for drug delivery†
Abstract
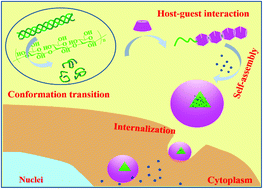
The unique conformation transition from a triple helix to single coils for the triple helical β-D-glucans has paved the way to fabricate various functional nanocomposites through the denaturing–renaturing process. This study firstly reports a novel kind of naturally derived supramolecular polymer micelle consisting of single-stranded chains of curdlan (CUR) and β-CDs. It is proposed that β-CDs as the host molecules were threaded onto single β-glucan chains (denatured triplex CUR) via the host–guest interaction, thereby forming supramolecular micelles. The results from the 1H NMR, FT-IR, XRD and 2D 1H NOESY NMR studies confirmed the formation of the inclusion complex and the existence of the core–shell structure of the supramolecular assembly. TEM images and DLS revealed that the self-organized micelles displayed a regular spherical shape with an average diameter of ∼27 nm. Furthermore, the hydrophobic anticancer drug camptothecin (CPT) was selected as a model drug and successfully encapsulated into the CUR/β-CD micelles. The drug-loaded micelles exhibited a steady sustained-release pattern regardless of the environmental pH. The flow cytometry and confocal laser scanning microscopy measurements confirmed that the CPT-loaded micelles could be well internalized into HepG 2 cells and continuously release the drug molecules inside the tumor cells. Meanwhile, the in vivo experiments demonstrated that CPT-loaded micelles could effectively inhibit tumor growth in comparison to free drugs. This concept will give a favorable platform to construct intelligent drug delivery systems for potential use.


 Please wait while we load your content...
Please wait while we load your content...