CD44 targeted redox-triggered self-assembly with magnetic enhanced EPR effects for effective amplification of gambogic acid to treat triple-negative breast cancer†
Abstract
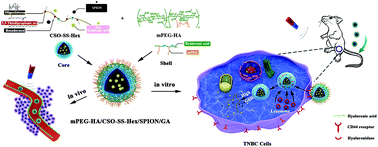
Gambogic acid (GA) is a natural anti-tumor drug whose application is restricted by its poor aqueous solubility and inefficient bioavailability. Developing nanomaterials with excellent biocompatibility can amplify the therapeutic effects of GA. In this study, a tumor-targeted redox controllable self-assembled nano-system with magnetic enhanced EPR effects (mPEG-HA/CSO-SS-Hex/SPION/GA) was developed to improve the anticancer efficacy of GA. The nano-system is constituted by three layers: the outer layer is mono-aminated poly(ethylene glycol) grafted hyaluronic acid (mPEG-HA), which can target the CD44 receptor in breast cancer cells; the middle layer consists of disulfide linked hexadecanol (Hex) and chitosan oligosaccharide (CSO) to control the drug release by reduction response; the core layer is superparamagnetic iron oxide nanoparticles (SPION), which can enhance the EPR effect by magnetic guidance and contribute to GA entrapment. Different experiments were performed to characterize the complex self-assembly, and the cytotoxicity, pharmacokinetics, and in vivo antitumor activity of the self-assembly were investigated to evaluate its anti-tumor effects. The results revealed that mPEG-HA/CSO-SS-Hex/SPION/GA is an excellent nanosystem with appropriate size and sensitive responsiveness; it can accumulate in tumor sites and achieve excellent therapeutic effects on triple-negative breast cancer (TNBC). In summary, a CD44-targeted redox-triggered self-assembly nanosystem with magnetic enhanced EPR effects was developed for effective amplification of GA; it has potential to act as an effective carrier in drug delivery for chemotherapy of TNBC.


 Please wait while we load your content...
Please wait while we load your content...