Fabrication of an ultra-sensitive para-nitrophenol sensor based on facile Zn-doped Er2O3 nanocomposites via an electrochemical approach
Abstract
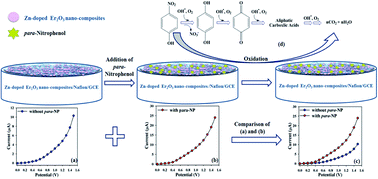
In this study, a semiconductor-doped nanocomposite material (Zn-doped Er2O3 nano-composites) was prepared via a single-step wet-chemical technique at alkaline pH. Fourier-transform infrared spectroscopy (FT-IR), UV/Vis spectroscopy, photoluminescence spectroscopy (PL), field emission scanning electron microscopy (FESEM), energy-dispersive X-ray spectroscopy (XEDS), and X-ray powder diffractometry (XRD) were applied to determine the structural and morphological properties of the Zn-doped Er2O3 nanocomposite. A thin layer of aggregated Zn-doped Er2O3 nanocomposite was fabricated on the flat surface of a glassy carbon electrode (GCE) with 5% ethanolic Nafion as conducting coating binder for the development of a selective and sensitive p-nitrophenol (para-NP) capturing electrochemical probe for environmental remediation. After the fabrication of the sensor, a novel current–potential (I–V) electrochemical approach was applied to determine its selectivity and sensitivity together with all the necessary analytical parameters against para-NP. Moreover, the calibration plot was found to be linear with the linear dynamic range (LDR) of para-NP concentration. The limit of detection (LOD) at a signal-to-noise ratio of 3 (S/N ∼ 3) and sensitivity were also calculated to be 0.033 ± 0.002 pM and 28.481 × 10−2 μA μM−1 cm−2, respectively, based on the gradient of the calibration plot, and the limit of quantification (LOQ) was determined to be 0.11 ± 0.02 pM. This work demonstrates a well-known approach for the first time that can be used for the development of efficient electrochemical sensors. These sensors based on semiconductor doped nanomaterials embedded onto the GCE for the detection of toxic chemicals in an aqueous system as an environmental remediation. It can be further applied for the analysis of real environmental samples and in the healthcare field.


 Please wait while we load your content...
Please wait while we load your content...