The investigation of molten ZnCl2 natural particle suspensions†
Abstract
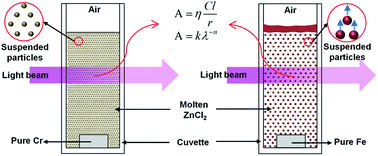
The common but unexpectedly neglected natural particle suspensions (NPSs) have significant effects on the characteristics of molten salts. In the present work, NPSs were generated via the corrosion of Cr or Fe cubes in molten ZnCl2 at 370, 420 or 470 °C in air and then investigated using in situ ultraviolet-visible spectroscopy, infrared spectroscopy and inductively coupled plasma optical emission spectrometry. The behaviors of Cr and Fe in these systems were quite different. In the case of Cr, particle (mainly contained Cr2O3) sizes grew over time and temperature. The elemental concentrations of Cr also increased over time; in addition, its values at 420 °C were slightly larger than that at 370 °C but much lower than that at 470 °C. For Fe, particle (mainly contained Fe2O3) sizes and elemental concentrations of Fe decreased with temperature, whereas they first increased (the time required for the increase reduced with temperature) and then decreased slowly over time. These results can be ascribed to the chemical reactions of these metals with the molten salts and/or various impurities.


 Please wait while we load your content...
Please wait while we load your content...