Assessment of ATR-FTIR spectroscopy with multivariate analysis to investigate the binding mechanisms of Ag and TiO2 nanoparticles to Chelex®-100 or Metsorb™ for the DGT technique†
Abstract
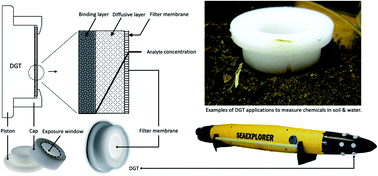
Studying nanomaterials' ecotoxicology is not new but there are still gaps in our understanding of their fate in the environment. A major challenge is lack of reliable tools to measure available concentrations of nanoparticles (NPs) in soil and water. Diffusive gradients in thin-films (DGT) is a robust technique for measuring the concentrations of trace metals in the environment. We have also shown that it could be potentially developed for measuring ZnO NPs. To further investigate the suitability of DGT for measuring the available concentrations of NPs in soil and water we selected two model nanoparticles, Ag and TiO2, which are widely used and incorporated in different commercial products. We aimed to understand (1) if two of the DGT binding agents, Chelex®-100 and Metsorb™, could irreversibly retain our model NPs and if yes (2) what might be the differences between bound Ag and TiO2 NPs and Ag+ and Ti4+ cations. We used ATR-FTIR spectroscopy for this purpose and analysed the IR spectra using principal component analysis and linear discriminant analysis (PCA-LDA), as our pattern recognition tool. The results show that the DGT resins form chemical bonds with silver and titanium nanoparticles and their ionic forms. PCA-LDA demonstrates that the binding mechanisms are statistically different (95% confidence level) among the treatments. This study indicates DGT's potential for measuring the available concentrations of NPs in the environment and suggests that ATR-FTIR spectroscopy combined with computational analysis could potentially differentiate between chemical species that are retained simultaneously by the DGT device resin layer.


 Please wait while we load your content...
Please wait while we load your content...