Isolation of extracellular vesicles with multivalent aptamers
Abstract
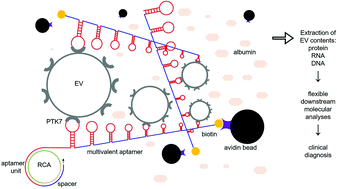
Extracellular vesicles (EVs) are lipid-enclosed submicron-sized vesicles that are secreted by all eukaryotic cells. EVs can selectively encapsulate tissue-specific small molecules from parent cells and efficiently deliver them to recipient cells. As signal mediators of intercellular communication, the molecules packaged in EVs play critical roles in the pathophysiology of diseases. In relevant clinical translation, EV contents have been used for cancer diagnosis and treatment monitoring. To further promote EV-based cancer liquid biopsy toward large-scale clinical implementation, the efficient and specific isolation of pure tumor-derived EVs from body fluids is a prerequisite. However, the existing EV isolation methods are unable to address certain technical challenges, such as lengthy procedures, low throughput, low specificity, heavy protein contamination, etc., and thus, new approaches for EV isolation are required. Here, we report a multivalent, long single-stranded aptamer with repeated units for EV enrichment and retrieval. After short incubation of biotin-labeled multivalent aptamers (MAs) with the samples, EVs can be quickly secured by MAs, anchored onto streptavidin-coated microspheres, and further retrieved via digestion of the DNA aptamer. Approximately 45% of EVs can be isolated from the spiked samples in 40 min with a depletion of 84.7% of albumin contamination. In addition, 93.1% of the isolated EVs can be retrieved via DNase-mediated aptamer degradation in 10 min for downstream molecular analyses. Our findings suggest that MAs can efficiently and specifically isolate EVs derived from malignant lymphocytes, and this simple method could facilitate the EV-centered study of acute lymphoblastic leukemia.


 Please wait while we load your content...
Please wait while we load your content...