Voltammetric pH sensor based on electrochemically modified pseudo-graphite†
Abstract
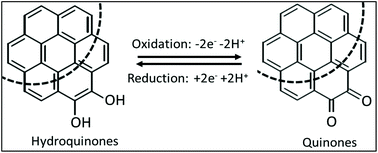
A nanocrystalline graphite-like amorphous carbon (graphite from the University of Idaho thermolyzed asphalt reaction, GUITAR) shares morphological features with classical graphites, including basal and edge planes (BP, EP). However, unlike graphites and other sp2-hybridized carbons, GUITAR has fast heterogenous electron transfer (HET) across its basal planes, and resistance to corrosion similar to sp3-C and boron-doped diamond electrodes. In this contribution, quinoid modified BP-GUITAR (q-GUITAR) is examined as a sensor for pH determination. This modification is performed by applying 2.0 V (vs. Ag/AgCl) for 150 seconds followed by 15 cyclic voltammetric scans from −0.7 to 1.0 V at 50 mV s−1 in 1.0 M H2SO4. The quinoid surface coverage of q-GUITAR is 1.35 × 10−9 mol cm−2, as measured by cyclic voltammetry. X-ray photoelectron spectroscopy analysis also confirms the high surface coverage. The quinoid surface concentration ranks highest in literature when compared with other basal plane graphitic materials. This yields a sensor that responds through a square wave voltammetric reduction peak shift of 63.3 mV per pH over a pH range from 0 to 11. The response on q-GUITAR is stable for >20 measurements and no surface re-activation is required between the measurements. The common interferents, Na+, K+ and dissolved oxygen, have no effect on the response of the q-GUITAR-based pH sensor.


 Please wait while we load your content...
Please wait while we load your content...