Detection of perfluorooctane sulfonate by ion-transfer stripping voltammetry at an array of microinterfaces between two immiscible electrolyte solutions†
Abstract
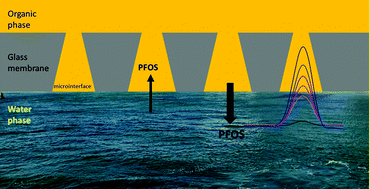
Per- and polyfluoroalkyl substances (PFAS) are a category of persistent environmental contaminants that have been linked to health issues in humans. In this work, we investigate the detection of perfluorooctanesulfonate (PFOS−), one such PFAS, by ion-transfer voltammetry at an array of microinterfaces between two immiscible electrolyte solutions (μITIES). Cyclic voltammetry, differential pulse voltammetry and differential pulse stripping voltammetry (DPSV) indicated the ion-transfer behaviour and detection of PFOS−, with the latter enabling detection at picomolar concentrations. Using a 5 min preconcentration time, during which PFOS− was preconcentrated into the organic phase of the μITIES array, a limit of detection (LOD) of 0.03 nM (0.015 μg L−1) in aqueous electrolyte was achieved. This performance is attributed to the enhanced mass transport (radial diffusion) to the μITIES that occurs during preconcentration. To investigate the potentiality for applications of this analytical approach to environmental samples, measurements in a range of water matrices were investigated. Drinking water, laboratory tap water and seawater matrices were assessed by spiking with PFOS− over the 0.1–1 nM range. A matrix effect was observed, with changes in sensitivity and LOD relative to those in pure aqueous electrolyte solutions. Such matrix effects need to be considered in designing applications of these PFOS− measurements to environmental samples. The results presented here indicate that DPSV at a μITIES array can form the basis for a fast and sensitive screening method for PFOS− contamination that is suited to portable and on-site applications.
- This article is part of the themed collection: Versatile Electrochemical Approaches


 Please wait while we load your content...
Please wait while we load your content...