New barbituric acid derivatives for data encryption and decryption based on the mechanochromic fluorescence effect†
Abstract
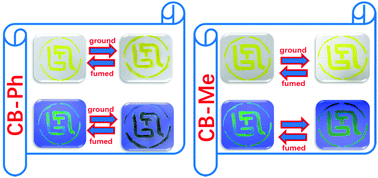
Two barbituric acid derivatives CB-Ph and CB-Me were synthesized, both of which show a strong aggregation induced emission (AIE) effect. It was found that these two compounds show almost the same absorption and emission spectra. As a result, they show the same yellow color in daylight and the same yellow-green color under UV at 365 nm. Upon grinding, CB-Ph exhibits superior mechanochromic fluorescence (MCF) properties, with its fluorescence color from yellow-green (555 nm) to brown (580 nm) and its emission intensity decreases by 93%. However, neither the fluorescence peak wavelength nor the intensity of CB-Me shows observable changes after being ground, indicating no MCF effect. The X-ray diffraction (XRD) data indicate that the CB-Ph powder changes from the crystalline to amorphous state after being ground, whereas CB-Me remains in its crystalline state. Molecular dynamics simulation (MDS) shows that CB-Ph takes a 3D conformation due to three phenyl groups on the periphery of the molecule, which hinders the molecules from aggregating closely and results in the change of the microstructure by external force stimuli. However, CB-Me aggregated more closely due to its better planarity and hydrophobic n-hexane group, which make it difficult to change the microstructure upon being ground. Based on the unique optical properties of CB-Ph and CB-Me, a new data encryption–decryption technology is developed. CB-Ph is used as cryptographic ink due to its excellent MCF effect upon grinding. CB-Me is used as a camouflage material as it shows the same color as CB-Ph in the daylight and under UV-365 nm; furthermore, its colors remain unchanged after being ground.


 Please wait while we load your content...
Please wait while we load your content...