A novel analytical principle using AP site-mediated T7 RNA polymerase transcription regulation for sensing uracil-DNA glycosylase activity †
Abstract
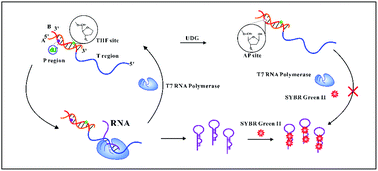
Uracil DNA glycosylase (UDG) is a highly conserved damage repair glycosylase; the abnormal expression of DNA glycosylase has important research value in many human diseases. Therefore, highly sensitive and specific detection of UDG activity is crucial to biomedical research and clinical diagnosis. In this work, we propose an AP site-mediated T7 RNA polymerase transcription regulation analytical principle for uracil-DNA glycosylase activity analysis. T7 RNA polymerase is highly promoter-specific and only transcribes DNA downstream of the T7 promoter. We have found that modifying the T7 promoter sequence with an AP site can regulate T7 RNA polymerase transcription ability according to different modification sites. In the binding region of the promoter, AP sites greatly inhibit transcription. Moreover, AP sites in the initiation region of the promoter enhance transcription activity. Based on this research, we designed a new transcription substrate template by replacing deoxythymidine (dT) in the T7 RNA polymerase promoter sequence with one tetrahydrofuran abasic site mimic (THF) and one deoxyuridine (dU). The THF site was labeled in the transcription-enhanced region to improve transcription background, and the dU site was labeled in the transcription inhibition region to sense the UDG enzyme. In our strategy, this template can be transcribed into RNAs by T7 RNA polymerase with great multicycle amplifications. When UDG is present, dU is excised to form an AP site. The AP site damages the interaction between T7 RNA polymerase and the T7 promoter, resulting in weak transcription activity. The detection limit of this strategy is as low as 2.5 × 10−4 U mL−1, and it has good selectivity for UDG. In addition, this strategy can also detect UDG activity in complex HeLa cell lysate samples. Therefore, our developed sensor might become a promising technique for UDG activity assay.


 Please wait while we load your content...
Please wait while we load your content...