Selective ratiometric red-emission detection of In3+ in aqueous solutions and in live cells using a fluorescent peptidyl probe and metal chelating agent†
Abstract
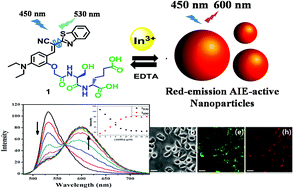
Indium has been regarded as one of the most rarely used metal ions; however, the consumption of indium has increased intensively due to its increasing use in electrodes of liquid crystal displays (LCDs). In recent years, warnings have been issued about the toxicity of indium to aquatic ecosystems and humans. Thus, the development of efficient and selective detection methods for In3+ in aquatic environments as well as in live cells is highly required. However, the selective and sensitive detection of In3+ in the presence of trivalent metal ions and other metal ions is highly challenging. In the present study, we synthesized a fluorescent probe (1) for In3+ and Al3+ based on an unnatural peptide receptor and an aggregation-induced emission fluorophore and developed a selective fluorescent detection method for In3+ in aqueous solutions and live cells using the probe and a metal chelating agent. 1 recognized In3+ and Al3+ selectively among 19 metal ions in aqueous solutions depending on pH by the enhancement of the red emission at 600 nm and decrease in the green emission at 530 nm. 1 sensitively detected In3+ and Al3+ by ratiometric response in a wide pH range (3.5–7.4), and the ratiometric response was complete within 20 seconds in an aqueous buffered solution at pH 5.0. Interestingly, the addition of EDTA to the complex of 1 with In3+ or Al3+ did not induce the Al3+-free spectrum but instead induced the In3+-free spectrum; thus, In3+ and Al3+ could be easily differentiated. The detection limit of 1 for In3+ ions was 211 nM (R2 = 0.981) in purely aqueous solutions. The fluorescence ratiometric detection method using 1 could quantify low concentrations of In3+ in ground water and tap water. Fluorescence cell image studies revealed that the probe was cell-permeable, and low concentrations of In3+ inside the cells could be recognized by the enhancement of the red emission at 600 nm. The binding mode study via NMR, IR, and CD spectroscopy revealed how the peptide receptor of 1 interacted with In3+ and resulted in the enhancement of the red emission in an aqueous solution.


 Please wait while we load your content...
Please wait while we load your content...