Electrochemiluminescence analysis of tryptophan in aqueous solutions based on its reaction with tetraphenylborate anions†
Abstract
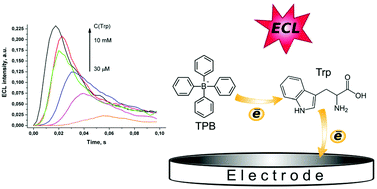
The emission of electrogenerated chemiluminescence (ECL) is observed upon the oxidation of fluorescent aromatic amino acid tryptophan in aqueous solutions at a glassy carbon electrode in the presence of a sodium tetraphenylborate co-reactant in a broad pH range. The emission is particularly strong when potential pulses are applied to the electrode. The ECL intensity is proportional to the concentration of amino acid and allows its determination in the concentration range of 0.3 μM–0.3 mM with high selectivity in the presence of other amino acids. The results of studies suggest that the indole ring of tryptophan is responsible for the observed ECL emission.


 Please wait while we load your content...
Please wait while we load your content...