Combined Raman and AFM detection of changes in HeLa cervical cancer cells induced by CeO2 nanoparticles – molecular and morphological perspectives†
Abstract
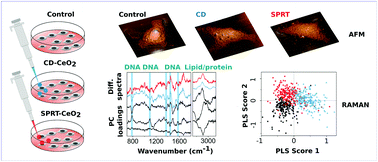
The design of nanoparticles for application in medical diagnostics and therapy requires a thorough understanding of various aspects of nanoparticle–cell interactions. In this work, two unconventional methods for the study of nanoparticle effects on cells, Raman spectroscopy and atomic force microscopy (AFM), were employed to track the molecular and morphological changes that are caused by the interaction between cervical carcinoma-derived HeLa cells and two types of cerium dioxide (CeO2) nanoparticles, ones with dextran coating and the others with no coating. Multivariate statistical analyses of Raman spectra, such as principal component analysis and partial least squares regression, were applied in order to extract the variations in the vibrational features of cell biomolecules and through them, the changes in biomolecular content and conformation. Both types of nanoparticles induced changes in DNA, lipid and protein contents of the cell and variations of the protein secondary structure, whereas dextran-coated CeO2 affected the cell-growth rate to a higher extent. Atomic force microscopy showed changes in cell roughness, cell height and nanoparticle effects on surface molecular layers. The method differentiated between the impact of dextran-coated and uncoated CeO2 nanoparticles with higher precision than performed viability tests. Due to the holistic approach provided by vibrational information on the overall cell content, accompanied by morphological modifications observed by high-resolution microscopy, this methodology offers a wider picture of nanoparticle-induced cell changes, in a label-free single-cell manner.


 Please wait while we load your content...
Please wait while we load your content...