Preparation of a chemically stable metal–organic framework and multi-walled carbon nanotube composite as a high-performance electrocatalyst for the detection of lead
Abstract
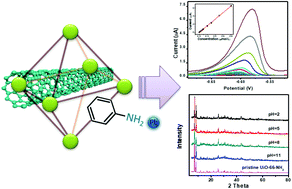
The fabrication of chemically stable metal–organic frameworks for electrochemical sensors is essential to broaden their applications. In this study, the integration of a Zr-based metal–organic framework (UiO-66-NH2) with multi-walled carbon nanotubes (MWCNTs) was achieved by a one-pot solvothermal reaction. This composite was then characterized by scanning electron microscopy, transmission electron microscopy, powder X-ray diffraction, Fourier transform infrared spectroscopy, and contact angle measurements. The porous structure and unreacted amine groups provided by UiO-66-NH2 frameworks accompanied by the excellent conductivity of MWCNTs have made this composite an ideal electrode material for lead detection. After the optimization of MWCNT content and the detection conditions, the oxidation peak currents were proportional to lead concentrations in the range of 0.001–0.8 μmol L−1 with a low detection limit of 0.5 nmol L−1 (S/N = 3). More importantly, the well-preserved crystallinity of MWCNTs@UiO-66-NH2 after 80 cycles and after being soaked in different pH aqueous solutions indicates the promising application of this sensor for real samples. Satisfactory recoveries were achieved when this sensor was used to detect lead in real samples. Furthermore, the excellent stability of this composite makes it possible to be employed in other fields, including extreme acidic media.


 Please wait while we load your content...
Please wait while we load your content...