Voltammetric detection of vitamin B1 (thiamine) in neutral solution at a glassy carbon electrode via in situ pH modulation
Abstract
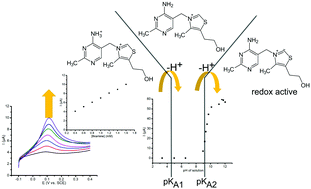
Voltammetric analysis is often dependent on pH and on the addition of buffer reagents to optimise the analytical procedure. This approach is not always possible for in situ analytical measurements, for example when studying biological fluids or ingredients in food. Therefore, a method is proposed herein, which employs a working electrode to do both, that is, to locally modulate the pH value and to measure the analytical response. As a model system, thiamine (vitamin B1) is detected in aqueous KCl with a pH modulation brought about with negative potentials applied to the working electrode. Interferences from ascorbic acid and uric acid are considered. Exploratory data are presented and methods for improving the detection limit are suggested. Their potential for applications in electroanalysis (and in a broader range of processes) is discussed and the detection of thiamine in rice is demonstrated.
- This article is part of the themed collection: Versatile Electrochemical Approaches


 Please wait while we load your content...
Please wait while we load your content...