One-step rapid synthesis of Ni6(C12H25S)12 nanoclusters for electrochemical sensing of ascorbic acid†
Abstract
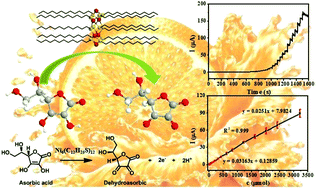
Metal nanoclusters (NCs) are highly desirable as active catalysts due to their highly active surface atoms. Among the reported metal clusters, nickel nanoclusters (Ni NCs) have been less well developed than others, such as gold, silver and copper. Herein a simple method is developed to synthesize atomically precise Ni clusters with the molecular formular of Ni6(C12H25S)12. Moreover, the single crystal of the Ni6(C12H25S)12 cluster is also obtained. The composition, morphology and optical properties of the prepared Ni6 clusters are characterized by X-ray crystallography, XPS, XRD, SEM, HRTEM, FTIR and UV-Vis spectroscopy. The Ni cluster is composed of six nickel atoms that form a hexagonal ring with an exterior 1-dodecanethiol shell, resembling a double crown. Meanwhile, the Ni6 NCs can be self-assembled into nanosheets due to their uniform size. It was found that the Ni6(C12H25S)12 clusters loaded on carbon black exhibit higher electrocatalytic activity than Ni nanoparticles towards ascorbic acid (AA) oxidation. The Ni6 clusters show high sensing performance for AA with a wide linear range (1–3212 μM) and a low detection limit of 0.1 μM (S/N = 3). The significantly enhanced catalytic activity can be ascribed to the high fraction of surface Ni atoms with low coordination in the sub-nanometer clusters. The present work not only provides a straightforward method for synthesizing atomically precise metal clusters but also indicates that ultrasmall Ni clusters can be used as highly efficient catalysts for the electrochemical detection of AA.
- This article is part of the themed collection: Versatile Electrochemical Approaches


 Please wait while we load your content...
Please wait while we load your content...