Retracted Article: Microelectrochemical cell arrays for whole-cell currents recording through ion channel proteins based on trans-electroporation approach†
Abstract
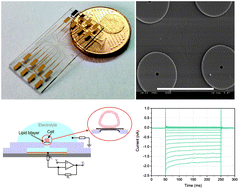
High electrostability and long life-time of planar chip technology are crucial for electrophysiological measurements such as ionic current recording through ion channel proteins embedded in biological cell membrane. In this paper, we propose a novel planar microchip integrated with microelectrochemical cell array toward to a feasible solution for ion channel screening with high resolution and long life-time. In order to reduce the interference from the leakage currents, a synthetic lipid bilayer is applied to form a high sealing resistance. The whole-cell electrical access can be constructed via electroporating the lipid bilayer in close proximity to the cell membrane. Parameters of electroporation including amplitude and time scale are firstly optimized by using parallel electroporation to the lipid bilayers. In this approach, individual cells can be trapped to the target positions by applying dielectrophoresis (DEP) manipulation. Poly(ethylene glycol) (PEG) is employed with low concentration to facilitate the closed contact between the cells and the lipid bilayer to increase the efficiency of the whole-cell mode construction. Through this chip based method, stable current recordings through inward rectifier potassium (Kir) ion channels embedded in rat basophilic leukemia (RBL-1) cell membrane are achieved with high electrical sealing resistance (over 1 GΩ). In addition, without need for complex fluidic connections, this method allows for an easy operation and further miniaturization of the measuring system.


 Please wait while we load your content...
Please wait while we load your content...