Investigating the structure–function relationship in triple cation perovskite nanocrystals for light-emitting diode applications†‡
Abstract
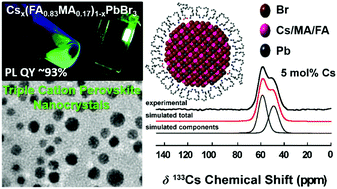
Organic metal halide perovskite nanocrystals are promising candidates for light-emitting diodes due to their narrow emission bandwidth, high photoluminescence quantum yield (PLQY), and color tunability. Nevertheless, these systems suffer from thermal instability, phase impurities, and a sensitivity to processing techniques. This study reports the first synthesis of novel Cs-containing triple cation perovskite nanocrystals with nominal stoichiometry Csx(MA0.17FA0.83)1−xPbBr3 (x = 0–0.15). The effect of Cs+ cation incorporation is thoroughly investigated using diffraction, microscopy and solid state MAS NMR techniques. The solid state 133Cs MAS NMR results reveals the distribution of the Cs+ cations is highly concentration and particle size dependent, with maximized surface/subsurface Cs+ concentrations being achieved with the smaller 5 mol% Cs system. These characteristics directly correlate improved surface passivation and environmental stability of the triple cation system. These triple cation nanocrystals exhibit a maximum photoluminescence quantum yield of ∼93% which upon translation to nanocrystalline LED devices delivers a maximum EQE of 7.4% (30 cd A−1) corresponding to a power efficiency of 34.87 lm W−1. This performance represents a marked improvement compared to CsPbBr3 nanocrystals (PL quantum yield ∼50%; maximum EQE of 2.5% (7.2 cd A−1)) fabricated under similar conditions.


 Please wait while we load your content...
Please wait while we load your content...