Polymorphism in N,N′-dialkyl-naphthalene diimides†
Abstract
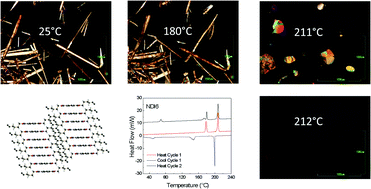
The long-known class of compounds called naphthalene diimides (NDI), bearing alkyl substituents on the imide nitrogen atoms, have been widely used as active materials in thin film devices with interesting optical, sensing and electrical applications. Less is known about their rich crystal chemical behaviour, which comprises numerous polymorphic transitions, and the appearance of elusive liquid crystalline phases. It is this behaviour which determines the response of the devices based on them. Here we fully characterized, by combining differential scanning calorimetry, powder and thin film diffraction and optical microscopy techniques, two newly synthesized NDI materials bearing n-octyl and n-decyl side-chains, as well as lighter analogues, of known room temperature crystal structures. In search for a rationale of their physico-chemical properties, phase stability and thermally induced solid-state transition reversibility, the differential behaviour of these NDI materials is interpreted here based on the competitive role of intermolecular π–π interactions and the alkyl chain flexibility. The appearance of comparable local minima of the molecular conformational energy hypersurface for shorter alkyls, and, for longer ones, of rotator phases, is here invoked.


 Please wait while we load your content...
Please wait while we load your content...